The role of IgG autoantibodies in CIDP
In patients with CIDP, the binding activity of autoantibodies, including IgG autoantibodies, to the myelin sheath may result in demyelination, subsequent nerve damage, and symptom expression.1,3,4
Infiltration of IgG autoantibodies
Activated T cells trigger inflammatory mediators (cytokines and chemokines) to break the blood-nerve barrier, facilitating the passage of autoantibodies, including IgG autoantibodies.1,2,4
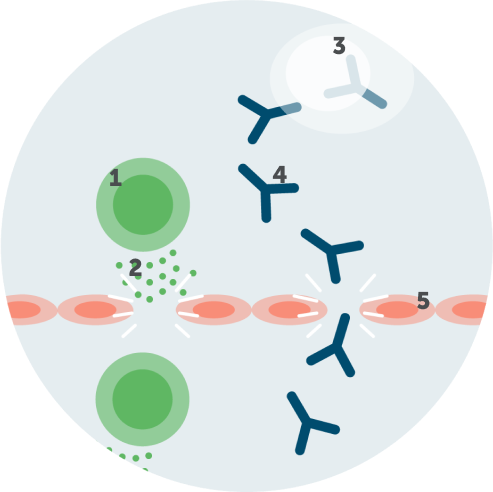
1. Activated T cell
2. Cytokines/chemokines
3. Plasma cell
4. IgG autoantibodies
5. Blood-nerve barrier
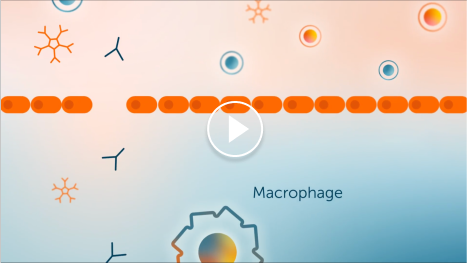
Demyelination due to IgG autoantibodies
IgG autoantibodies bind to the myelin sheath, where they either recruit macrophages directly or activate the complement pathway, which may result in myelin damage.1-4
1. IgG autoantibodies
2. Macrophage
3. Fc receptor
4. Myelin sheath
5. Complement proteins
Demyelination can contribute to axonal damage in CIDP and increase the potential for chronic disability.5-7
The GBS|CIDP Foundation estimates
30%
of patients with CIDP progress to
wheelchair dependence if not treated8
The GBS|CIDP Foundation estimates
30%
CIDP=chronic inflammatory demyelinating polyneuropathy; Fc=fragment, crystallized; GBS=Guillain-Barré syndrome; IgG=immunoglobulin G.
FcRn extends circulation of IgG antibodies, including autoantibodies
FcRn perpetuates the circulation of IgG antibodies, including the autoantibodies that may play a role in disease activity.5-7,9,10
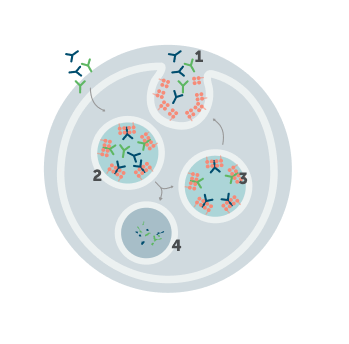
1. IgG antibody
2. Endosome
3. FcRn
4. Lysosome
Understanding how FcRn recycles IgG, including autoantibodies, opens new opportunities for intervention.9,10
FcRn=neonatal Fc receptor; IgG=immunoglobulin G.
You’ve explored the role FcRn plays in CIDP. Now learn about what
happens when it becomes a therapeutic target
CIDP=chronic inflammatory demyelinating polyneuropathy; FcRn=neonatal Fc receptor; MOA=mechanism of action.
References: 1. Mathey EK et al. J Neurol Neurosurg Psychiatry. 2015;86(9):973-985. doi:10.1136/jnnp-2014-309697 2. Querol LA et al. Neurotherapeutics. 2022;19(3):864-873. doi:10.1007/s13311-022-01221-y 3. Dziadkowiak E et al. Int J Mol Sci. 2022;23:2-13. doi:10.3390/ijms23010179 4. Koike H et al. Neurol Ther. 2020;9:213-227. doi:10.1007/s40120-020-00190-8 5. Ricciardi D et al. Brain Sci. 2022;12(11):1510. doi:10.3390/brainsci12111510 6. Al-Zuhairy A et al. Clin Neurophysiol. 2021;132(4):1000-1007. doi:10.1016/j.clinph.2020.12.017 7. Al-Zuhairy A et al. Muscle Nerve. 2022;66(6):715-722. doi:10.1002/mus.27722 8. GBS CIDP Organisation Europe. CIDP. Accessed January 9, 2026. https://eu.gbs-cidp.org/about/cidp/ 9. Ulrichts P et al. J Clin Invest. 2018;128(10):4372-4386. doi:10.1172/JCI97911 10. Wolfe GI et al. J Neurol Sci. 2021;430:118074. doi:10.1016/j.jns.2021.118074